With Anewex R & D, you can do it!
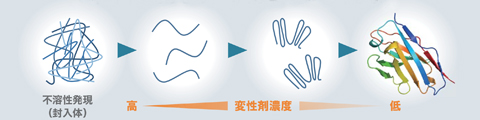
- I want to make a protein in E. coli that cannot be soluble and expressed in E. coli!
(If you do not have a facility such as an animal cell expression system, but want to produce protein yourself) - We accept consideration of rewinding conditions from insoluble fractions.